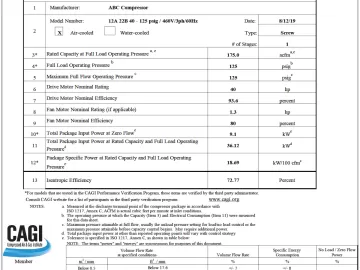
Isentropic Efficiency of Rotary Screw Air Compressors
Many astute air compressor users have noticed the Compressed Air and Gas Institute (CAGI) air compressor data sheets, dated after June 2020, have a new term listed; isentropic efficiency. Isentropic Efficiency will be the new standard of reference for a true comparison of the overall efficiency of air compressors at any rated discharge pressure. Now users can see which company produces the most efficient product with an easy reference percentage number. The compressed air industry, in conjunction with CAGI, has been trying to make fair comparisons between air compressors for years.