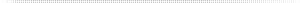This article will examine in detail four of the five acceptable WAGD implementations under NFPA 99, along with some alternative ways they may be implemented. This article will not deal with passive implementations.

 |
| BeaconMedaes AGS System |
Dual-Use Medical Vacuum/WAGD Implementations
Dual-use implementations are primarily medical vacuum systems into which the WAGD is introduced, more or less as a “free rider” on the medical vacuum network and pump. Dual-use implementations are high vacuum. The medical vacuum application will always take precedence when setting pump cut-in and cut-out. This means the lowest vacuum a WAGD inlet can see is 12 inHgV (305 mmHgV per NFPA requirements for medical vacuum) and typical vacuums are 15–29 inHgV (380-760 mmHgV). Due to the high vacuum, the networks for these systems must be relatively strong and copper pipe is therefore commonly used. WAGD terminals look very similar to vacuum inlets except for color, and are nearly identical to vacuum inlets internally. Most significantly, this means that the internal porting is limited, and many WAGD inlets (particularly older ones) may have some difficulty with higher flows. Piping is typically sized using the same loss tables as are used for vacuum, and valving and alarming generally follows the vacuum rules. A rare variant on this implementation, is a regulated WAGD line. In these implementations, a regulator is installed in the WAGD line, which reduces the top vacuum level to a medium or low vacuum and thus brings the WAGD pipeline closer to the ideal of an intrinsically safe system. Such an implementation requires separate sizing of the WAGD line at the lower vacuum. Copper pipe would still be the preferred material of construction.
Dedicated WAGD, Using a Pump
Pump-based implementations have in the past generally been run at vacuum levels similar to medical vacuum. Pumps usually selected are designed to run at vacuums of 15 inHgV (380 mmHgV) or higher. However, a variant of this system is a medium to low vacuum system, wherein the systems operate more nearly in line with the ideal of an intrinsically safe system. This is achieved by sizing and setting the pumps to lower vacuum settings (e.g. 5-10 inHgV {127-250 mmHgV}) and sometimes also installing a vacuum regulator in line to reduce the vacuum at the inlet. This variant is appropriate only with technologies that are suited to low vacuum operation (liquid ring, dry vane, rotary lobe).
The networks must be designed to handle the full vacuum of which the pump is capable (even though the pump may in fact operate at a lower vacuum normally). Therefore, the networks for these systems must be relatively strong. Copper pipe is most commonly used. At vacuums 12 inHgV {305 mmHgV} and above, piping would be sized using the same loss tables as are used for medical vacuum, but alternative sizing methods will be required at lower vacuums. WAGD terminals used at the upper end of the vacuum scale (5 inHgV and higher) look very similar to vacuum inlets, except for color, and are nearly identical to vacuum inlets internally. Most significantly, this means that the internal porting is limited, and many WAGD inlets (particularly older ones) may have some difficulty with higher flows. Therefore, at the lower end of the vacuum scale, the terminals may need to be of a different type to ensure adequate flow.
Dedicated WAGD, Using a Blower
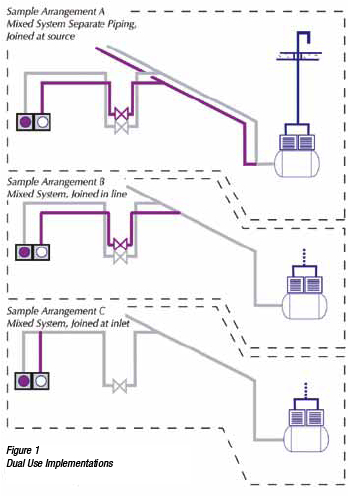
These implementations achieve all the objectives desired in a WAGD implementation, but typically are the most complex to design and install. Unlike the two types discussed previously, these systems typically use a regenerative blower as their producer. Whereas a pump is designed to expand the air first (i.e. produce a deep vacuum) and move volume second, a blower is designed to move volume first and produce only a shallow vacuum. In this they more closely resemble a fan than a pump. It is this emphasis on moving lots of air at low vacuums that makes them ideal for WAGD.
As an example, a liquid ring pump with a one horsepower motor will move 396 lpm (14 scfm) at 5 inHgV (if it could be operated there — most will run up to 28 inHgV). A regenerative blower driven with the same one hp motor will move 1,245 lpm (44 cfm) at 40 inH2OV (2.9 inHgV). This low vacuum creates it’s own issues. The sizing of the network is immediately different from that used for higher vacuum, and the terminals must be of a different type to pull enough flow at these low vacuums.
However, an important advantage of these low vacuum implementations is that the producer may be located close to the terminals, unlike pumps, which typically must be located remotely. A blower especially a small one) is sufficiently compact and quiet that it can sometimes be placed near the WAGD terminals (e.g., in a ceiling space, a mop closet, etc.) The network can therefore be minimized. A pump, being typically larger and noisier, must typically be placed at some remove in a mechanical space. The resulting network is longer and potentially more complex (see Figure 2).
 At first glance, it may appear to be more expensive to implement and operate a local system possibly composed of multiple blowers vs. a single large pump. In fact, because blowers are less expensive, smaller and internally less complex than pumps of similar capacities, the economics often slant in favor of the local implementation.
At first glance, it may appear to be more expensive to implement and operate a local system possibly composed of multiple blowers vs. a single large pump. In fact, because blowers are less expensive, smaller and internally less complex than pumps of similar capacities, the economics often slant in favor of the local implementation.
Low vacuum systems can be piped in a variety of materials. NFPA requires they be metallic and non-corroding, which rules out plastic or iron pipe. However, copper, stainless and galvanized pipe might be used, as might ductwork and thin-wall galvanized because of the low vacuum (an ideal material would seem to be electrical conduit with liquid tight fittings, but so simple an answer may be too exotic to be acceptable in many local jurisdictions). Copper pipe is the most common material. These systems may require a means to balance the system and are tested in a somewhat different manner to high vacuum systems.
WAGD Using Distributed Producers
These implementations are in many ways the simplest, but have not been seen in the North American market and are thus unfamiliar. They tend to be relatively expensive to install, because two separate piping networks are required.
Conceptually, they involve no central producer, but instead use a venturi in each inlet. The venturi is intrinsically capable only of low vacuum. An air line must serve the venturi, and medical air is not permitted for use in this service. Instrument air is ideal, but at this writing relatively few facilities have instrument air systems. Once the venturi is served by an appropriate air source, the exhaust must also be routed to the outside and sized. The exhaust side may be made of pipe suitable to the pressure. Copper would be typical. There are no alarms, which can practically be installed. Each inlet is individually controlled, and must have an operating indicator of its own.
All WAGD implementations, except the distributed producer styles, share the same basic requirements as to the location of terminals, alarms, etc. All WAGD implementations share the same basic requirements as to the discharge from the building. Specific details of how these items operate will vary by implementation. We will consider the universal requirements here, and then deal with the necessary variations under each specific implementation.
WAGD Selection
What factors should be weighed when selecting a WAGD implementation? There are several, and the weighting to be given to each will vary from facility to facility. They include:
1. Effectiveness. Will the system do the job of keeping the workspace free of waste gas? Efficacy usually has less to do with the type of system selected than the design and installation of the system, as all the implementations described herein are perfectly capable of being effective if well designed.
2. Patient safety. Will the system protect the patient and ensure the anesthesiologist’s control of the procedure? Here, the low vacuum implementations are to be preferred over the high vacuum implementations due to the intrinsic safety implied in a lower vacuum.
3. Cost. Which system is least expensive to implement and operate? Evaluating this is complex and the result varies dramatically between facilities. In general, low vacuum systems are less expensive than are high vacuum systems. Low vacuum systems are also typically lower maintenance than are high vacuum systems.
However, an assertion often made in favor of dual-use implementations is that WAGD dumped into a medical vacuum system is “free”, since the medical vacuum “has to be there anyway”. When the average WAGD inlet only flowed 6–9 liters, there were many cases where this was at least in part true. With WAGD flowing at 50 liters (1.8 scfm), it is true far less often. The additional capacity required and the additional operating hours mean that the cost of WAGD produced by a medical vacuum pump is considerably higher than has been assumed.
A simple rule of thumb test can be applied: Size the medical vacuum system without WAGD and select a pump of appropriate capacity. Add in the WAGD requirement (use at least 1.8 scfm per location). If the pump selected has sufficient capacity to handle the additional volume, an argument can be made that the WAGD produced is “free”, or at least low cost. If the pump selected does not have the necessary excess capacity, and thus to accommodate the WAGD a larger pump must be selected, a properly selected dedicated system will almost certainly be less expensive. This is especially true when a low vacuum system is used for comparison. Remember that a horsepower of pump will move approximately 15 scfm, whereas a horsepower of blower will move approximately 44 scfm — a ratio of roughly 3:1.
4. Technology. Is the technology otherwise preferred for the medical vacuum source acceptable for WAGD? If not, can another option be equally acceptable? In some cases, a technology otherwise preferred for use with medical vacuum may be oxygen sensitive, and there is not an equally acceptable oxygen-compatible alternative. Naturally, this will restrict the potential for dual use. The same limitation may also render unacceptable pumps otherwise preferred in a dedicated pumped system. In such a case, the limited technology options may be a powerful argument in favor of a low vacuum alternative.
5. Design complexity. How difficult is the system to design and what are the chances of problems resulting from bad design? Whatever can be said against a dual-use system, they are undoubtedly among the simplest to design. Correspondingly, low vacuum dedicated systems offer the greatest range of advantages for the user, but are probably the most complex to design and are also outside the experience of most North American designers. Low vacuum dedicated systems are also the most complex to commission.
WAGD Design: General Requirements
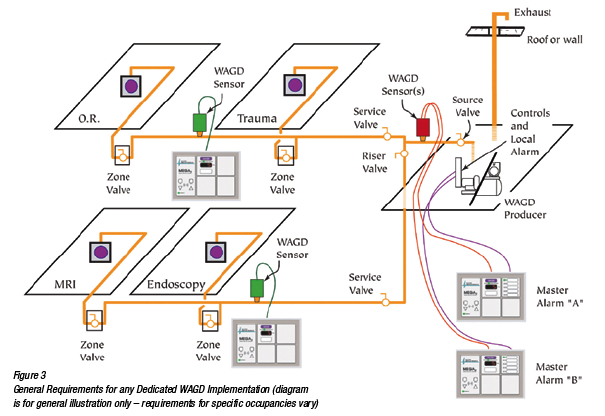
(Reference Figure 3)
NFPA 99 states that a unique, dedicated WAGD terminal should be placed wherever nitrous oxide or halogenated anesthetic is intended to be administered (13.3.5.2, 14.3.5.2). This will obviously include any location piped with nitrous oxide. Consideration should also be given to areas that are not traditionally piped with nitrous, but where nitrous oxide mounted on the anesthetizing machine can reasonably be expected to be used. Common examples of these locations include CAT/MRI/PET scan rooms, lithotripsy, exam/treatment rooms, trauma rooms and other locations where patients are routinely sedated or anesthetized. Other areas that should receive consideration might include laboratories where veterinary anesthesia will occur, dental clinics and OB/Labor where nitrous oxide/oxygen self-administration is practiced. WAGD terminals may also need to be placed in areas such as recovery, where exhaled anesthesia from recovering patients must be considered and the staff protected.
All dedicated WAGD producers are required to be duplex, wherein one unit must be sufficient to serve the system, and a second of equal size is ready to operate in the event of any inadequacy in the first. A local alarm indicating Lag WAGD producer in service must be included and relayed to the master alarm. The WAGD producer is required to include a source valve. Exhausts from the producer must exit the building, discharging at least 10 feet from any opening in the building, at a vertical level different from any air intake (preferably at a lower level) and in a location that is open and permits free dispersion of the waste gas.
Electrical power must be from the essential electrical system, equipment branch. Centrally piped WAGD is required to be valved like any other medical gas or vacuum system. Valves may be either ball or butterfly type. Required valves include: source valve, main valve (in a limited number of circumstances), riser valves, service valves and zone valves.
WAGD inlets must be separate from and non-interchangeable with the vacuum inlets (even if they are ultimately piped to the same source). WAGD has it’s own color code (white letters on violet).
Many older WAGD or evacuation inlets were “one-way” interchangeable (you could plug vacuum into evacuation, but not evacuation into vacuum). This is no longer permitted and such inlets should be retrofitted to bring them up to standard. The master alarms for a piped WAGD system will include at least an indicator for “Low WAGD” and an indicator for “WAGD Lag Producer Running.”
Any area fitted with piped WAGD requires a WAGD area alarm at the nurses’ station, just like any other medical gas. It will typically be piped into the line upstream of the anesthetizing location zone valve.
Distributed WAGD may be considered exempt from some of these requirements, but others may need to be fulfilled in unusual ways. As an example, while it is impractical to alarm a venturi, it is appropriate to alarm the drive air so that the facility knows if the WAGD is inoperable. It is necessary to observe intent in these cases and to ensure the essential functions are present even if it is necessary to use different methods to achieve the result.
For more information, please contact Mark Allen, BeaconMedæs, email: mark.allen@beaconmedaes.com, tel: 704-588-0854, www.beaconmedaes.com.
To read medical industry articles, visit www.airbestpractices.com/industries/medical.