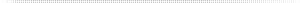Introduction
Compressed air is an often underestimated but crucial utility, the quality of which must be monitored periodically to fulfill national and international standards. The International Organization for Standardization (ISO) publishes world-class standards for products, services and systems, to ensure quality, safety and efficiency (1). ISO 8573 is an available standard addressing compressed air quality. It consists of nine parts that address purity classes, specifications, and procedures. ISO 8573-7:2003, the most current version, can be utilized across all industries’ compressed air microbial monitoring plans. It contains both informative and normative procedures but lacks any tested compressed air microbial specifications regarding colony enumeration limits for microbial plate counts. Specifications do exist for the required blind samples. This article will focus on ISO8573-7 normative test methods and analysis for viable microbiological contaminants and how it can be fundamentally utilized in compressed air microbial monitoring plans.
|
About the Author Maria Sandoval has over 15 years of experience in Microbiology and Molecular Biology. Her field work includes analyzing extremophiles isolated from the depths of Lake Baikal in Russia to the 50km exclusion zone of Chernobyl. Additionally, she’s worked alongside the CDC with DSHS analyzing and diagnosing patient microflora. Her tenure with the Lawrence Berkeley National Laboratory, Department of State Health Services and the University of Texas MD Anderson Cancer Center has made her a leading expert in microbial testing. As Trace Analytics’ Microbiologist, she is responsible for microbial testing and procedural development. |

Maria Sandoval, Microbiologist, Trace Analytics |
| Trace Analytics is an A2LA accredited laboratory specializing in compressed air and gas testing for food and beverage manufacturing facilities. Using ISO 8573 sampling and analytical methods, their laboratory tests for particles (0.5-5 microns), water, oil aerosol, oil vapor, and microbial contaminants found in compressed air. For over 29 years, they’ve upheld the highest industry standards of health and safety, delivering uncompromising quality worldwide in accordance with ISO, SQF, BRC, and FDA requirements. Visit www.AirCheckLab.com. | |
Choosing a Method that Fits your Needs
Analyzing micro burden data at point of use outlets throughout compressed air pipeline systems at a given time, acts as a window of observation into the control of the facility. Maintaining control means proper preventative maintenance, microbial monitoring scheduling and risk assessment must be appropriate for the industry being monitored. Many accreditation bodies can aid in the understanding of microbial limits and specifications, critical to specific industry needs. Once the compressed air microbial monitoring plan is approved, a sampling procedure that provides the company with the results suitable to its limits and specifications needs to be established. This requires the use of a procedure that accurately measures and samples a specific volume of air for microbial burden analysis inside the tested compressed air system.
Searching on the internet today, one can find a plethora of options, ranging from small, sleek portable samplers to robust pieces of equipment like the SAS Pinocchio Super II sampler. It is imperative when choosing the piece of equipment for testing that the technician and monitoring supervisor read the original equipment manufacturer (OEM) manual to establish what the limits and restrictions are for that product. For instance, while the portable samplers are lightweight and easily transportable, the OEM recommendations may state (2), “Due to the air flow pattern inside the unit, it is not recommended that CFUs get counted. Rather, the results should be viewed qualitatively.” As it stands, this instrument, though ideal for qualitative binary results, does not provide useful information for total plate counts mandated by ISO 8573-7 or any monitoring plan requiring validated colony forming unit enumeration.
Some microbial air impact samplers, like the SAS Pinocchio Super II, are designed to diffuse the air flow from a regulated point of use outlet, and then measure the flow rate to achieve the recommended rate by the OEM. This allows the end user to only monitor the time required to meet the desired total sample air volume; in most cases, 1000 liters. When using impact samplers, various sampling heads exist to disperse the air onto the recipient agar plate. Slit samplers and sieve samplers are the most prevalent.
The most common question about sieve samplers is whether to include the corrections adjustment for the post analysis reporting. If a sieve sampler is used, note the correction table (Table 1) from SAS 180 Microbial Air Samplers, which is extrapolated from the Macher article referenced below (3,4). Since the SAS Pinocchio Super II uses the same sieve head as the SAS 180 environmental air sampler head, the adjustment can be used. The correction is made to the final colony forming unit count that corresponds to the number of holes inside the sampling head relative to the plate size. The correction adjustment can be utilized on behalf of all perforated sampling heads with 219-hole, 401-hole, or 487-hole impactor for the statistical possibility of multiple particles passing through the same hole on to the agarose surface. If the facility’s microbial limitations are under the need for the corrections adjustment, then the point is moot.
ISO 8573-7 Requires Two Blinds and a Sterility Blank
ISO 8573-7 dictates that two types of controls exist that “shall be” free from contamination. The first type of control plates is called blinds, these blinds (before and after) are to confirm that no outside or unwanted contamination from external variables (sampling technicians and the environment) have entered the air sampler or agarose surface. It should be noted that lack of aseptic technique can cause an entire sampling point to fail, resulting in unplanned costs and retesting. The blind is taken with the same movements as a compressed air sample, but no air is to enter the funnel head or impact the agarose. Two blinds are taken, one before the point of use and one after. The second type of control plate required, is a negative control, or sterility blank.
Compressed Air Purification & Piping Monthly e-NewsletterWith a focus on Demand-Side Optimization, compressed air dryers, filters, condensate management, tanks, piping and pneumatic technologies are profiled. How to ensure system reliability, while reducing pressure drop and demand, is explored through System Assessment case studies. |
There is no name in the ISO standard for this control plate, however, there is a description as follows: “Using the same means of transport, “geographically” trace a Petri dish the entire distance from the manufacturer who filled the Petri dishes with agar, to the place of sampling and the laboratory, in order that it can be inspected for unintended after-contamination. The dish shall not subsequently show growth.” This plate’s sole purpose is to travel the length of its existence, unopened, from cradle to grave, until its final analysis in the testing lab. Both controls must be <1 CFU/plate to be considered a “pass” from an ISO 8573-7 stand point. These are the only viable microbial specifications and limitations in ISO 8573-7:2003.
Taking a Compressed Air Sample Is Easy!
Most impact air samplers are made to use a sterile tube to sample the outlet air into the inlet port of the unit for analysis. For simplicity reasons this article will focus on the use of a Pinocchio Super II sampler. The compressed air is then passed through a manifold that measures the air through the calibrated flowmeter, and pressure gauge. Per the OEM the compressed air must measure to an appropriate rate (in this case 100LPM) with pressure under 3 Bar. Once the flow rate has been established, the air valve to the pressure gauge and flowmeter is closed. Since no adjustments were made at the inlet valve, the valve to the sampling funnel head can now be completely opened for measurement.
See Table 2 for calculations of measurement times relative to air flow rate. When the appropriate time has lapsed to sample 1000 liters, the inlet valve is closed, the plate is removed aseptically, labeled, and shipped to the testing lab for analysis. When sampling media is placed on the clips for testing, all efforts should be made to avoid secondary contamination. It only takes one breath, sneeze, loose gloved finger to make an entire point of use sample worthless. Paying attention to aseptic technique is imperative to taking a true compressed air microbial sample.

Why Is Aseptic Technique so Important?
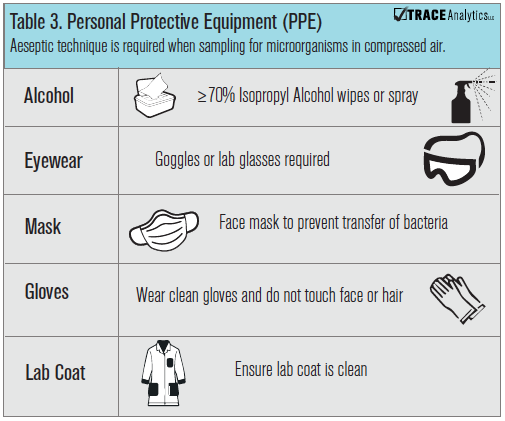
Aseptic technique is a term that refers to movements, actions, attire and attitude when working in a scientific or environmental sampling capacity. Testing compressed air may not seem like the proverbial environmental sample, but it is. Taking a small sample of the microbial profile of a point of use environment and growing it up in a laboratory takes skill and awareness. Aseptic technique functions to prevent contamination of media and reagents by microorganisms. The first action is to always wear the minimum appropriate personal protective equipment, PPE (Table 3): fitted gloves, eye protection, clean lab coat, and hair net. Before handling any media, gloves should be rubbed down or sprayed with greater than or equal to 70% Isopropanol Alcohol or Ethanol. Not doing this important step will almost certainly result in contaminated blinds and samples. When in doubt, change your gloves or wipe them down with alcohol. The simplest and most economical way to reduce contamination is to work on a cleaned, disinfected work area.
Food industries who sample on sorting floors are subjected to a lot of dust. Cleaning and disinfecting the area that the microbial air sampler sits upon should be routine and thorough. Afterwards, open the contact plate or petri dish for sampling, the lid should always be placed down, as to not catch any floating microbial vectors like dust inside the lid. These are all points of contamination that are easily avoidable. Most plates are sent with sterile bags to send back to the testing lab. Feel free to place the lid face down inside the sterile bag provided, making sure gloves are clean.
Sampling technicians should make sure to never open multiple petri dishes at a time. Sealing the petri dishes is the next best mode of avoiding contamination. If a locking mechanism exists on the plate, make certain to lock it once the sample is taken. Always parafilm or tape the plate, this protects the lid from falling off during transit, and keeps moisture in the plate. Good personal hygiene acts to not only protect the sampling technician from environmental contaminants, but also protects the samples from contamination like shed skin, dirt and dust from street clothes.

Understanding the Results of an ISO 8573-7 Report
Once the final microbial report has been issued, certain criteria must be met to adhere to ISO 8573-7. Mentioned above, the before and after blinds, must be reported as <1 CFU/plate. The distinction between CFU/plate and CFU/m3 is made when no volume of air is tested and when 1000L of air is tested, respectively. The sterility blank must also be free of contamination and reported as <1 CFU/plate. The compressed air sample should fall inside of the facility’s monitoring plan limits and specifications. Remember to always keep in mind of the measurement of uncertainty that ISO 17025 labs report.
Laboratory measurements always involve uncertainty, which must be considered when analytical results are used as a part of a basis for making decisions like pass/fail. Some monitoring plans require that the actual sample number fall within the limitations of the measurement of uncertainty. Usually it is the responsibility of the facility, not the lab to determine a pass/fail status.
The only analysis required by ISO 8573-7 is colony forming unit enumeration. However, if a facility requires microbial identification, Gram staining is done for bacterial colonies. Gram staining classifies most bacterial colonies into two groups; Gram positive or Gram negative. Additional information is noted on shape and morphology upon Gram staining. Most pathogenic organisms can begin to be identified from this simple strain. For instance, if the air sample was contaminated with Gram positive cocci bacteria, there is no need to test for Listeria since Listeria is a Gram positive, non-spore forming rod. It should be noted however, that these samples, while informative, are a snap shot of the level of control in the facilities compressed air system. Continual monitoring and quarterly or at minimum biannual testing is recommended.
Safety and Quality in Compressed Air: Why You Should Care – Webinar RecordingDownload the slides and watch the recording of the FREE webcast to learn:
|
Summary
- ISO 8573-7:2003 is the leading standard for testing compressed air.
- ISO 8573-7 requires before and after blinds, one sterility blank and absolutely no contamination on them.
- ISO 8573-7 requires a validated impact air sampler for colony forming unit enumeration, read the OEM manual before renting or purchasing to confirm.
- Aseptic technique is required, it will save you money if you learn it, and cost you money if you don’t.
- ISO 8573-7 only requires colony forming unit enumeration.
- Gram stain differentiation is an easy way to begin the process of pathogenic identification.
References
- All about International Organization for Standardization. https://www.iso.org/about-us.html
- Compressed Air Microbial Testing Unit Detection Kit [Manual]. (07/2015)
- SAS 180 Air Sampler [Manual]. (03/2007)
- Macher, J. (1989). Positive-Hole Correction of Multiple-Jet Impactors for Collecting Viable Microorganisms. American Industrial Hygiene Association Journal, 5(11), 561-568.
For more information please contact Trace Analytics at tel: 800-247-1024, email: ServiceTeam@AirCheckLab.com or visit www.AirCheckLab.com.
To read similar articles on Compressed Air Standards please visit www.airbestpractices.com/standards/iso-cagi.