Compressed air and gases are vital to numerous healthcare facility operations. Commonly used for breathing, sedation, and the operation of medical instruments, healthcare facilities must rely on these utilities for lifesaving and therapeutic benefits. The quality of the air and gas produced by the facility’s compressed air systems is paramount to their efficacy in promoting positive outcomes for patients.
Contamination of compressed air/gas, whether from ambient air, the air compressor itself, or the piping system, is a liability for healthcare facilities that can threaten patient health and safety, and ultimately cost the facility in malpractice lawsuits.
In 1996, at least four patients died and 70 were injured due to the liver-damaging effects of trichloroethylene, a solvent commonly used to clean piping and gas tanks, contamination in their bulk oxygen tanks1. Hospital staff had noticed an odor shortly after tank changeover1, a clear signal of contamination that should not have been ignored and that happens to be part of air/gas criteria per National Fire Protection Association (NFPA) Health Care Facilities Code ² (https://www.nfpa.org/codes-and-standards/all-codes-and-standards/list-of-codes-and-standards/detail?code=99).
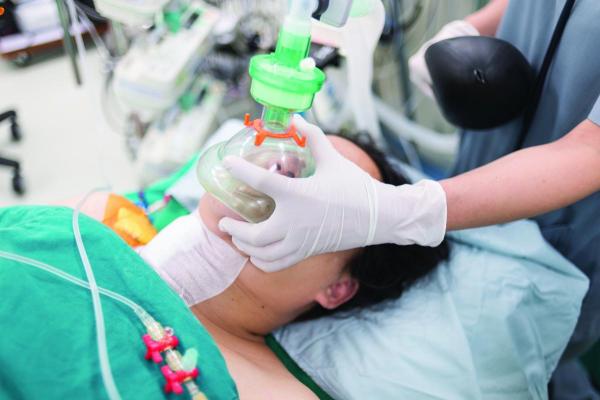
The quality of the air and gas produced by compressed air systems at healthcare facilities is paramount to their efficacy in promoting positive outcomes for patients.
NFPA 99 – A Closer Look
Regulatory agencies, including the Centers for Medicare and Medicaid Services (CMS), The Joint Commission (TJC), and local authorities having jurisdiction (AHJ), mandate that healthcare facilities ensure their compressed air and gas systems are compliant with NFPA 99.
NFPA 99 lists odor, water, carbon monoxide, carbon dioxide, gaseous hydrocarbons, halogenated hydrocarbons, oil and particulates (non-viable and viable) as major contaminants to monitor and sets concentration purity requirements for medical gases as outlined in Table 1.
Kyle Jussel, experienced and certified medical gas system verifier and president of the Medical Gas Professional Healthcare Organization (MGPHO), notes that, “NFPA 99 requires that as verifiers, we perform initial testing [for these contaminants] at the source as well as each point of use. Facilities are also required to show that every accessible medical gas outlet/inlet has been tested and/or inspected according to their own risk assessment with consideration of the equipment manufacturer’s recommendations, which is typically on an annual basis thereafter.”
Note that NFPA 99 does not include specific criteria for viable particulates, such as bacteria, mold, and yeast. However, it may be of critical importance for healthcare facilities to verify their compressed air and gas is free of potential microbial contamination.
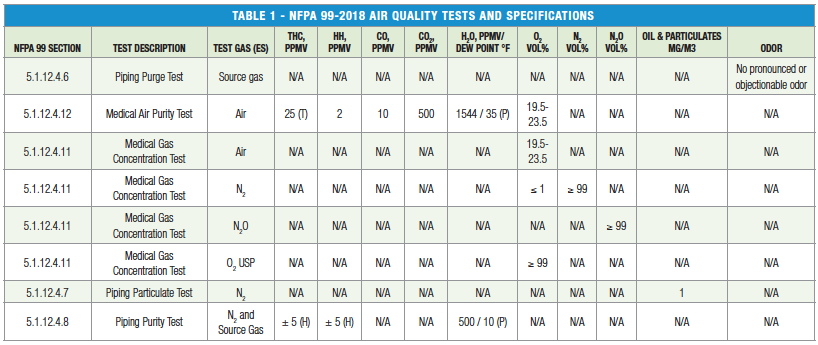
THC= Total Gaseous Hydrocarbons, CO= Carbon Monoxide, CO2= Carbon Dioxide, H2O= Water, O2= Oxygen, N2= Nitrogen, N2O= Nitrous Oxide
(T) Includes Methane
(H) Excludes Methane, difference allowed between nitrogen and source gas
(P) Referenced at 50 psi
Understanding Regulated Contaminants
Before delving into adverse implications of contamination, it is important to understand how contamination can occur. According to Jussel, “One of the major sources of contamination that we typically see when verifying medical gas systems includes copper particulate, which usually results from installers that may not have done proper purging during their initial testing procedures. We also occasionally see evidence of burnt pipeline plugs that were never removed prior to installation. This can be easily found during the odor test that is required of verifiers per NFPA 99.”
Contamination, however, is an issue not just observed during installation and verification. Jussel explains that, “Once a system has been properly installed, verified and placed into service for patient use, we have seen problems with high levels of CO, CO2 and humidity/dewpoint. All of which can result from maintenance related issues and/or poor intake air quality.”
Understanding of NFPA 99 regulated contaminants and their potential adverse effects is essential to not only complying with NFPA 99 codes for air/gas criteria but also to ensuring the quality of medical gases used in healthcare facilities once compliance has already been met. Examples are shown in Table 2.
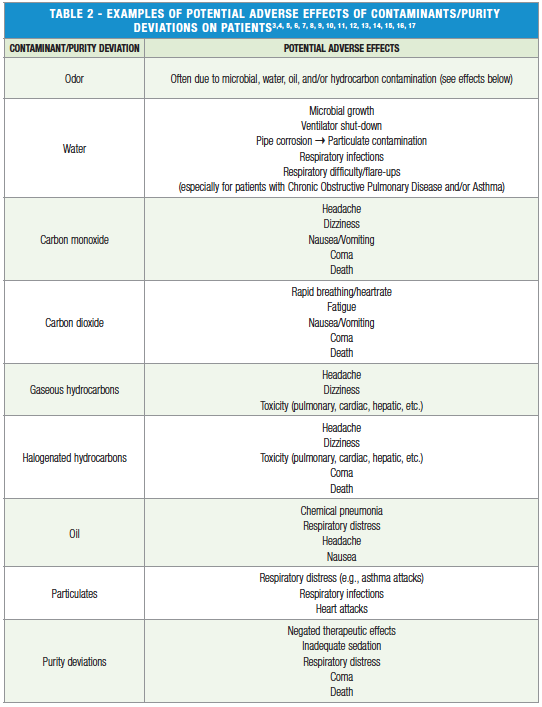
It begs reiteration, as noted in Table 2, that contamination and/or deviations in gas purity can lead to life-threatening injuries for patients. NFPA 99 only stipulates extensive air/gas verification for newly installed, repaired, or modified compressed air and gas systems and/or based on the air compressor manufacturer’s recommendations2. Compressed air and gas systems that do not meet these criteria, such as previously verified and/or older systems, may be exempt from additional verification per NFPA 99. NFPA 99 does, however, require air quality monitors, though monitoring may not include and/or separately identify all contaminants tested for during verification, such as halogenated hydrocarbons like trichloroethylene. Thus, it is to the benefit of healthcare facilities to continuously monitor and test their compressed air and gas systems in order to protect their patients, achieve high compressed air system efficiency to save money, and provide evidence in legal matters.
Compressed Air and Gas Verification Testing
To ensure compliance with NFPA 99, healthcare facilities must complete piping purge, piping particulate, piping purity, medical gas purity, and medical gas concentration verification testing (See Table 2).
NFPA 99 requires testing/analysis be performed by a party other than the installer2. And when a system is not installed by in-house personnel, testing can be performed by an in-house American Society of Safety Engineer (ASSE) 6030-qualified employee2. ASSE 6030 qualified employees can complete verification testing per NFPA 99 using portable analyzers/monitors or in conjunction with a third-party laboratory. This gives healthcare facilities the ability to save money and be proactive in minimizing patient and facility risk.
There are advantages to both portable analyzer and third- party laboratory testing methods. Portable analyzers, for example, may provide quicker results and in real-time. However, laboratory testing is often of higher accuracy and quality due to more complex techniques, instrumentation, and data analysis. Thus, a testing laboratory can sometimes provide clues as to the source of contamination through description/identification of particles, hydrocarbons, oils, etc.
If using a laboratory for analysis, it is in the best interests of healthcare facilities to only use accredited testing laboratories, such as Trace Analytics, LLC, as this ensures analysts, testing equipment, methods, quality assurance, and testing data meet or exceed internationally accepted standards (e.g., ISO 17025 for testing laboratories).
Jussel remarks that, “By sending air samples in for third-party laboratory analysis, we are able to give our customers peace of mind knowing that the quality of the air that they are delivering to their patients has been tested in a redundant fashion.”
Jussel goes on to explain that, “Not only was the medical air system tested upon start-up/verification, but it is also being supervised on a continuous basis by the air quality monitors required by NFPA 99. The results of which can also be compared to the third-party laboratory analysis that we provide on an annual basis.”
In addition to providing objective assurance for healthcare facilities, third-party laboratory testing may also be cheaper in the long run. Portable analyzers can be expensive if maintained by the healthcare facility themselves, requiring calibration and frequent maintenance. Ultimately, healthcare facilities should determine their verification and continuous testing needs based on facility-specific risk assessments.

Microscopic analysis of particulates aids in troubleshooting contaminants.
Utilization of Proper Air Treatments
Utilization of proper air treatments is crucial to meeting air/gas quality requirements set forth by NFPA 99.
To combat water contamination, desiccant dryers are commonly used in healthcare facilities to remove water from the compressed air/gas as NFPA 99 requires dryers be designed to provide air at a maximum dewpoint of less than 32 °F at 50-55 psi2. While refrigerated dryers are effective dryers for many applications, they often cannot provide air below a 35 °F dewpoint, especially at low-flow conditions, which are common in hospitals, as condensate is not removed as effectively by the centrifugal separator. Dryer systems are often redundant, with NFPA 99 requiring duplexed systems, to prevent water contamination in the event of a malfunction. Desiccant dryers can release particulates into the compressed air system as desiccant dust is created as a result of the constant movement/friction of the desiccant as air flows through. This illustrates how the compressed air system and air treatments themselves may produce contaminants which must be continually monitored for and controlled.
A series of filters can be used to remove particulates, originating either from the ambient air or the air compressor itself. It is important to note that a point-of-use filter is vital to ensuring that the compressed air/gas is particle-free upon use. Condensed oil, another contaminant analyzed for NFPA 99 verification testing, can originate within the air compressor, when oil lubricated systems are employed, and/or from the ambient air. To remove condensed oil, coalescing filters can be employed. For gaseous contaminants, such as volatile hydrocarbons and carbon dioxide, activated charcoal filters/towers and catalytic converters can be employed, respectively.
Air treatments not only remove contamination, but they also allow a compressed air system to work more efficiently, saving money by prolonging air compressor life and allowing the system to work faster. Employing air treatments and performing air compressor maintenance, in combination with compressed air/gas testing, allows healthcare facilities to ensure the quality of this utility and therapeutic.
Testing Frequency and Trend Analysis
NFPA 99 requires verification testing for new, modified, or repaired compressed air systems and subsequent testing per manufacturer’s recommendations, often annually. However, this testing frequency may not be sufficient to ensure compress air/gas quality year-round. Some facilities may opt to test quarterly as seasonal changes can alter the quality of the compressed air/gas. For example, high water concentrations are more common in the summer than in the winter.¹â· Other facilities may test before and after performing compressor maintenance, to ensure contamination was not introduced as a result of the repairs.
It is important to understand that testing only provides data for the compressed air system at the moment of sampling. To best optimize testing to ensure compressed air/gas quality, trend analysis can be employed. Trend analysis can utilize air quality monitoring data, obtained from monitors required per NFPA 99, in conjunction with third-party laboratory testing data to provide a detailed picture of air quality over time. Compressed air systems are dynamic – changes in seasons, system wear and tear, and other factors can all impact air/gas quality. Gathering sufficient data over time accounts for seasonal and maintenance changes and allows healthcare facilities to establish baseline quality levels for contaminants. Facilities can then best determine air compressor maintenance/cleaning schedules, potentially saving facilities time and money. In addition to establishing a baseline, healthcare facilities can set action and alert levels, which can allow the facility to anticipate contamination issues before they set off monitor alarms and/or threaten patient safety.

Reviewing data graphically can illuminate important trends.
Ensure Patient Safety with Testing and Monitoring
NFPA 99 Health Care Facilities Code is an important standard for ensuring the quality and safety of compressed air and gases used in healthcare facilities. Deviations in air purity and/or contamination could jeopardize patient safety. To comply with NFPA 99 and protect patients, healthcare facilities should employ air treatments and perform routine compressed air/gas testing and monitoring.
About Trace Analytics, LLC.
Trace Analytics, LLC is an ISO 17025 accredited laboratory specializing in compressed air and gas testing for healthcare facilities. Using validated sampling and analytical methods, their laboratory tests for air and gas purity, particles (0.5-5 microns), water, gaseous contaminants (e.g., carbon monoxide, carbon dioxide, total hydrocarbon content, and halogenated hydrocarbons), oil aerosol, oil vapor, and microbial contaminants. For over 30 years, they’ve upheld the highest industry standards of health and safety, delivering uncompromising quality worldwide in accordance with NFPA 99 requirements. Trace Analytics, LLC also works with experienced service distributors, such as Medical Air Testing & Services, Inc., to allow for easy, reliable, and affordable testing. For more information, visit www.airchecklab.com.
All photos courtesy of Trace Analytics.
To read similar NFPA 99 Medical Air Standards articles, please visit: www.airbestpractices.com/standards/nfpa-99-medical-air.
References
1Moss, E. and Evans, F. Hospital Deaths. Anesthesia Patient Safety Foundation. Retrieved July 27, 2019 from https://www.apsf.org/article/hospital-deaths/
²NFPA 99 Healthcare Facilities Code (2018)
³Mould, damp and the lungs. European Respiratory Society. Retrieved July 28, 2019 from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5709805/
â´Allen, E. E. (2018). Medical Gas System Safety. The Doctor’s Company. Retrieved July 20, 2019 from https://www.thedoctors.com/articles/medical-gas-system-safety/
âµMoss, E. Medical Air. Anesthesia Patient Safety Foundation. Retrieved July 21, 2019 from https://www.apsf.org/article/medical-air/
â¶Carbon Monoxide Poisoning. Centers for Disease Control and Prevention. Retrieve July 19, 2019 from https://ephtracking.cdc.gov/showCoRisk.action
â·Pietro, M. (2018). Humidity levels and COPD. Medical News Today. Retrieved July 21, 2019 from https://www.medicalnewstoday.com/articles/323657.php
â¸Carbon Dioxide OSH Answers Fact Sheets. (2019). Canadian Centre for Occupational Health and Safety. Retrieved August 1, 2019 from https://www.ccohs.ca/oshanswers/chemicals/chem_profiles/carbon_dioxide.html
â¹Toluene OSH Answers Fact Sheets. (2019). Canadian Centre for Occupational Health and Safety. Retrieved August 1, 2019 from https://www.ccohs.ca/oshanswers/chemicals/chem_profiles/toluene.html
¹â°Trichloroethylene OSH Answers Fact Sheets. (2019). Canadian Centre for Occupational Health and Safety. Retrieved August 1, 2019 from https://www.ccohs.ca/oshanswers/chemicals/chem_profiles/trichloroethylene.html
¹¹1Brown, K. W. and Armstrong, T. J. (2019). Hydrocarbon Inhalation. StatPearls. Retrieved July 21, 2019 from https://www.ncbi.nlm.nih.gov/books/NBK470289/
¹²Breathing Air Quality, Sampling and Testing. Environmental Health Laboratory, department of Environmental and Occupational Health Sciences, School of Public Health, University of Washington. Retrieved July 28, 2019 from https://deohs.washington.edu/sites/default/files/images-envhlth/topic/BreathingAirQualitySamplingandTesting_Technical.pdf
¹³Francis, J. Breathing Gas Contamination. Divers Alert Network. Retrieved August 1, 2019 from https://www.diversalertnetwork.org/health/PSA-GasContamination/index.html
¹â´Particle Pollution and Your Health. U.S. Environmental Protection Agency. Retrieved July 28, 2019 from https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P1001EX6.txt
¹âµHealth and Environmental Effects of Particulate Matter (PM). U.S. Environmental Protection Agency. Retrieved July 27, 2019 from https://www.epa.gov/pm-pollution/health-and-environmental-effects-particulate-matter-pm
¹â¶Moss, E. Medical Gas Contamination An Unrecognized Patient Danger. Anesthesia Patient Safety Foundation. Retrieved August 2, 2019 from https://www.apsf.org/article/medical-gas-contamination-an-unrecognized-patient-danger/
¹â·Water Vapor. Trace Analytics, LLC. Retrieved August 2, 2019 from https://www.airchecklab.com/aircheck-academy/breathing-air/sources-of-contamination/water-vapo