Air Quality in the Pharmaceutical Industry
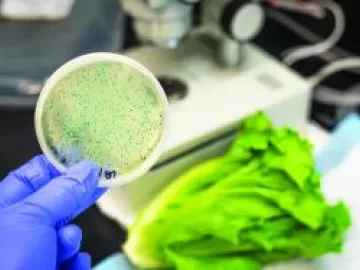
The United States accounts for roughly half of the global pharmaceutical market. This certainly keeps the Food and Drug Administration (FDA) busy in its oversight of pharmaceutical safety and effectiveness, including with the production processes. As the pharmaceutical industry has grown, so too has its utilization of compressed air for breathing air, operation of equipment and instrument air.