CAGI Personnel Certification: How to Have Your Own Personal Performance Verification Program
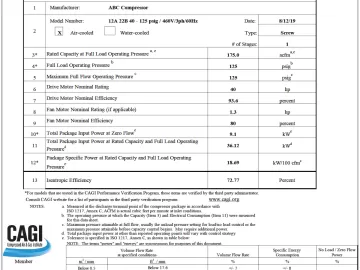
CAGI sponsors and oversees a performance verification program for some types of equipment. In this program, which is expanding to include additional types of products, a third-party verifies performance data that is stated by manufacturers. Participants in these programs are able to use a standardized CAGI datasheet to state performance. The datasheets and verification process provide customers with assurance that they are getting what they pay for in terms of performance.